In world first UK approves gene therapy for sickle cell disease: Basics Explained

Recently, the United Kingdom became the first country to approve the use of the world’s first gene therapy, is based on the CRISPR gene editing tool, for treating sickle cell disease and a specific type of sickle cell disease called beta-thalassemia. Globally, the current remedy for sickle cell anaemia is limited to bone marrow transplants.
Sickle cell disease — an inherited blood disorder that causes the body’s red blood cells to become misshapen and not last as long as healthy cells. Sickle cell disease affects the red blood cells in the blood, which are responsible for carrying hemoglobin throughout the body. Hemoglobin is a protein that binds to oxygen and delivers it to the tissues and organs throughout the body. This can lead to anaemia.
LEARNING FROM HOME/ WITHOUT CLASSES/ BASICS
A gene is the basic physical and functional unit of heredity. Genes are made up of DNA. Some genes act as instructions to make molecules called proteins. However, many genes do not code for proteins. Every person has two copies of each gene, one inherited from each parent.
DNA, or deoxyribonucleic acid, is the hereditary material in humans and almost all other organisms. Nearly every cell in a person’s body has the same DNA. Most DNA is located in the cell nucleus (where it is called nuclear DNA), but a small amount of DNA can also be found in the mitochondria. Watson and Crick discover chemical structure of DNA.
Gene therapy is a technique that uses a gene(s) to treat, prevent or cure a disease or medical disorder. Often, gene therapy works by adding new copies of a gene that is broken, or by replacing a defective or missing gene in a patient’s cells with a healthy version of that gene. Both inherited genetic diseases (e.g., hemophilia and sickle cell disease) and acquired disorders (e.g., leukemia) have been treated with gene therapy.
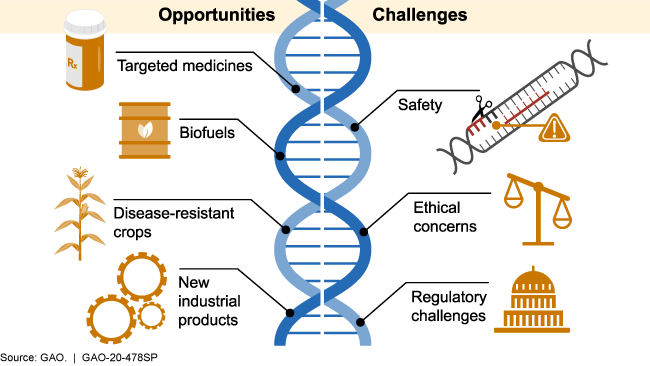
Genome editing employs variants of site directed nuclease (SDN) technologies and oligonucleotide-directed mutagenesis (ODM). Clustered Regularly Interspaced Short Palindromic Repeat/CRISPR associated proteins (CRISPR/Cas) systems are currently the most prominent SDN approaches.
Ethical concerns arise when genome editing, using technologies such as CRISPR-Cas9, is used to alter human genomes. Most of the changes introduced with genome editing are limited to somatic cells, which are cells other than egg and sperm cells (germline cells). These changes are isolated to only certain tissues and are not passed from one generation to the next.
However, changes made to genes in egg or sperm cells or to the genes of an embryo could be passed to future generations. Germline cell and embryo genome editing bring up a number of ethical challenges, including whether it would be permissible to use this technology to enhance normal human traits (such as height or intelligence). Based on concerns about ethics and safety, germline cell and embryo genome editing are currently illegal in the United States and many other countries.





0 Comments